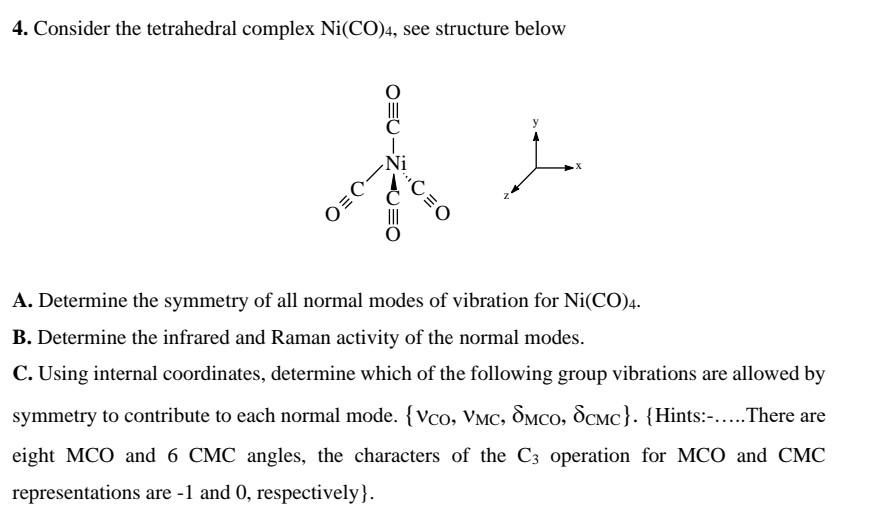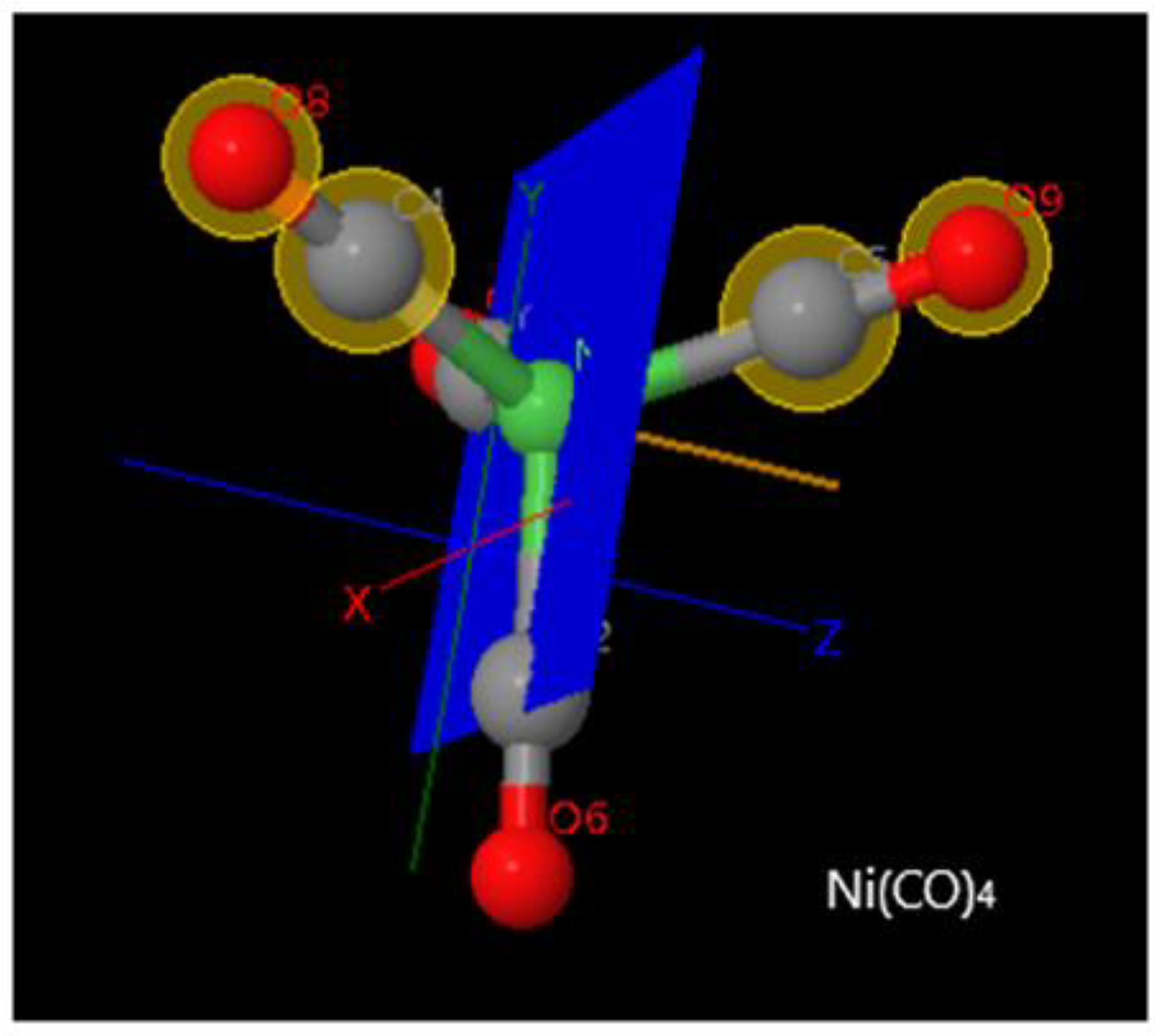
Chemistry | Free Full-Text | Dissociative Electron Attachment Cross Sections for Ni(CO)4, Co(CO)3NO, Cr(CO)6

The reactions of Cr(CO)6, Fe(CO)5, and Ni(CO)4 with O2 yield viable oxo‐metal carbonyls - Sun - 2014 - Journal of Computational Chemistry - Wiley Online Library
![Write IUPAC names of following compounds: (b) Ni(CO)4 (c) K3[Fe(CN)6] (e) Na2[ZnCl4] (1) K4[Fe(CN)6] (a) [Cr(en)3] C13 (d) [Co(NH3)4C12]CI (g) K3[Al(C2O4)3] 6) (Coſen)2Cl2]* (m) Na3[Fe(C204)3] (h) (Ag(NH3)2]C1 (k) [Ni(en)3]3+ (n) Na3[Co(NO2)6] ( Write IUPAC names of following compounds: (b) Ni(CO)4 (c) K3[Fe(CN)6] (e) Na2[ZnCl4] (1) K4[Fe(CN)6] (a) [Cr(en)3] C13 (d) [Co(NH3)4C12]CI (g) K3[Al(C2O4)3] 6) (Coſen)2Cl2]* (m) Na3[Fe(C204)3] (h) (Ag(NH3)2]C1 (k) [Ni(en)3]3+ (n) Na3[Co(NO2)6] (](https://toppr-doubts-media.s3.amazonaws.com/images/10134247/7fd28e88-c796-45e8-91b4-d623bd01bf03.jpg)
Write IUPAC names of following compounds: (b) Ni(CO)4 (c) K3[Fe(CN)6] (e) Na2[ZnCl4] (1) K4[Fe(CN)6] (a) [Cr(en)3] C13 (d) [Co(NH3)4C12]CI (g) K3[Al(C2O4)3] 6) (Coſen)2Cl2]* (m) Na3[Fe(C204)3] (h) (Ag(NH3)2]C1 (k) [Ni(en)3]3+ (n) Na3[Co(NO2)6] (
![The geometry of \\[Ni{{(CO)}_{4}}\\]and \\[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\\]are :(A) Both square planar(B) Tetrahedral and square planar, respectively(C) Both tetrahedral(D) Square planar and tetrahedral, respectively The geometry of \\[Ni{{(CO)}_{4}}\\]and \\[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\\]are :(A) Both square planar(B) Tetrahedral and square planar, respectively(C) Both tetrahedral(D) Square planar and tetrahedral, respectively](https://www.vedantu.com/question-sets/26080d1f-f0b7-482a-b3ca-4dc1b6ce1eb64722761873556963438.png)
The geometry of \\[Ni{{(CO)}_{4}}\\]and \\[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\\]are :(A) Both square planar(B) Tetrahedral and square planar, respectively(C) Both tetrahedral(D) Square planar and tetrahedral, respectively
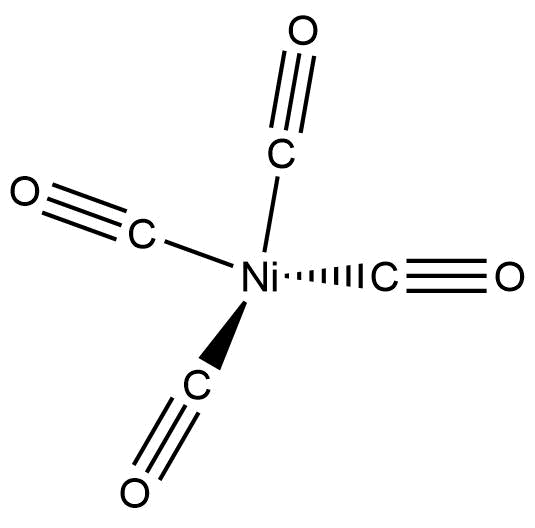
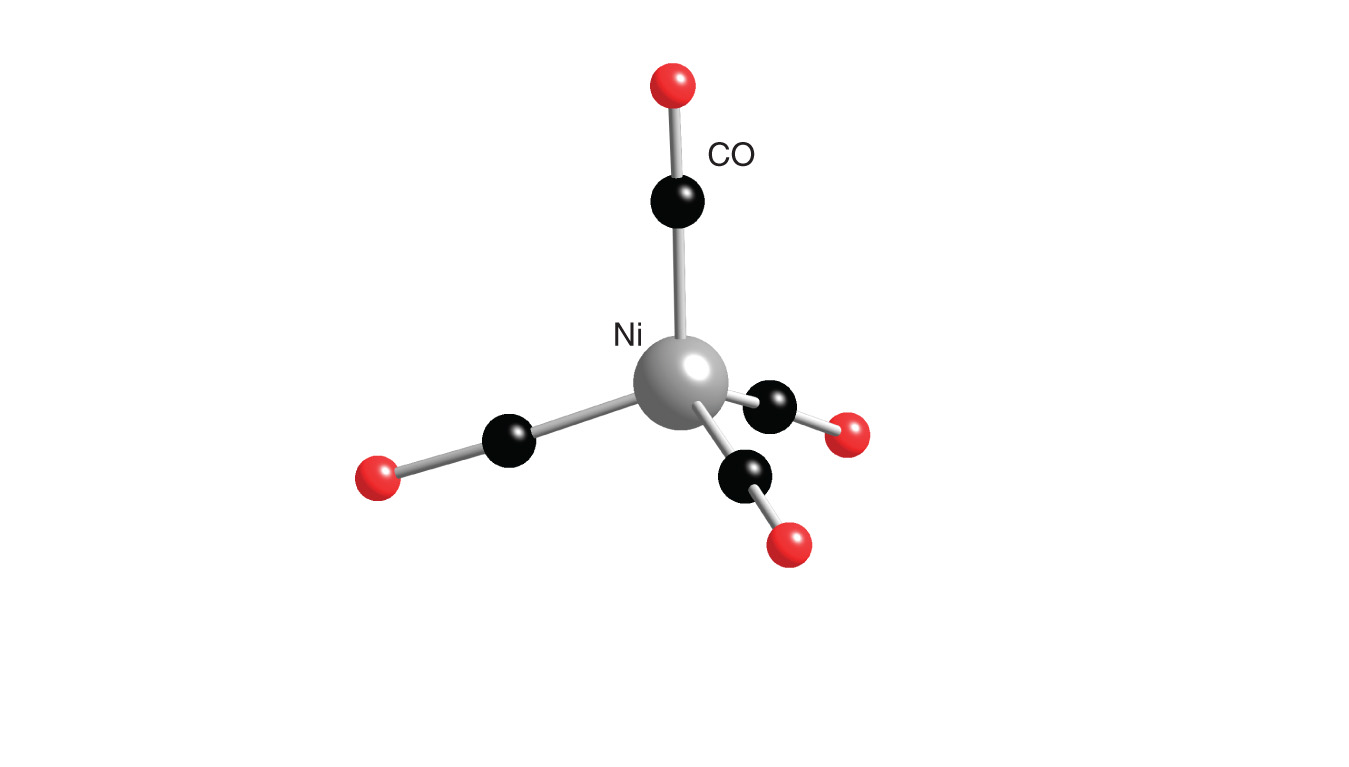
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-1.png)
![Ni(CO)_(4)]` का IUPAC नाम लिखे- - YouTube Ni(CO)_(4)]` का IUPAC नाम लिखे- - YouTube](https://i.ytimg.com/vi/hAYjZvomAXU/maxresdefault.jpg)
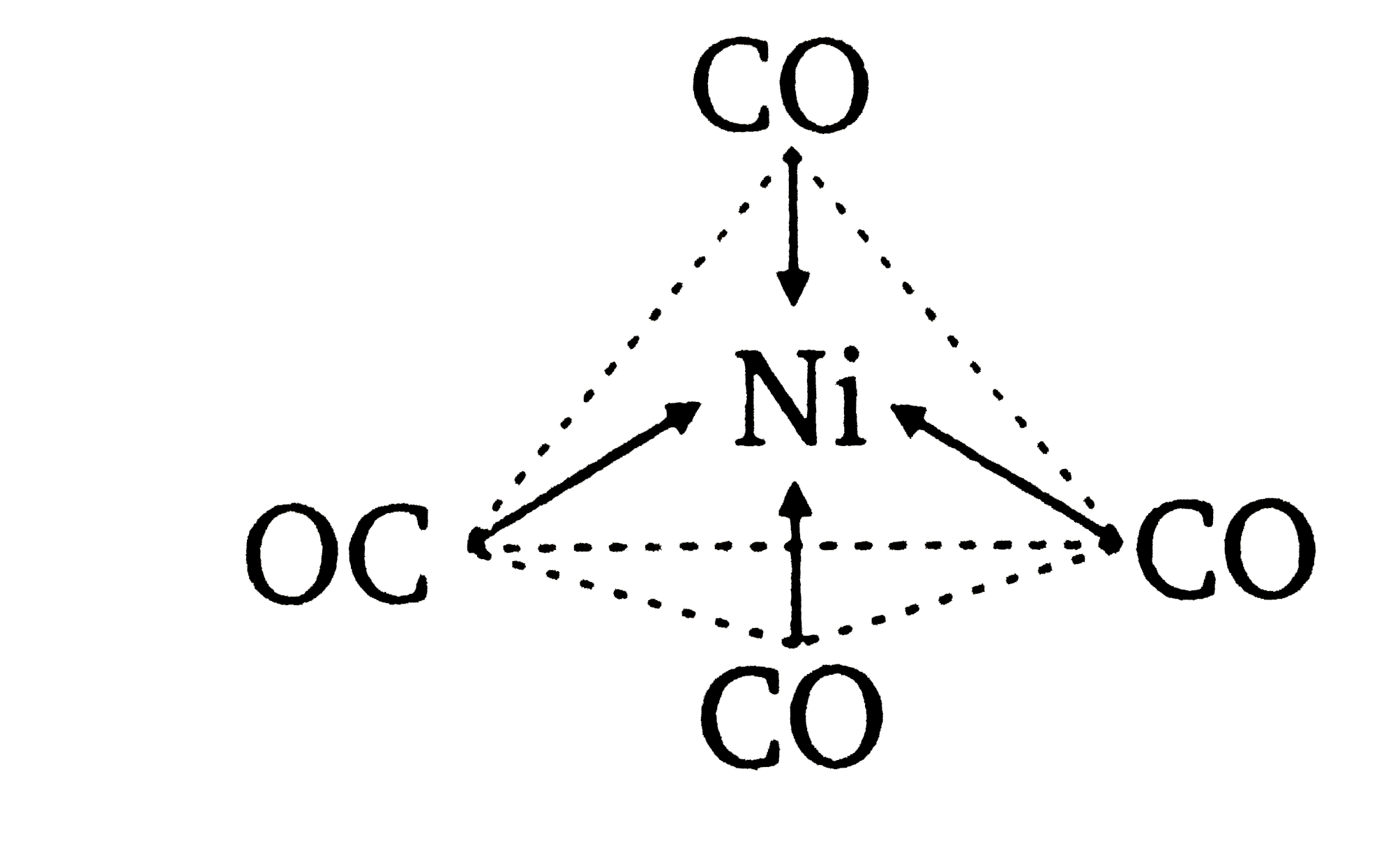


![Ni(CO)4 has a tetrahedral geometry whereas [Ni(CN)4]4&ndash Ni(CO)4 has a tetrahedral geometry whereas [Ni(CN)4]4&ndash](https://www.zigya.com/application/uploads/images/chen12070385_571483de917be.png?t=1460962273442)
4.png)
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-2.png)



![The geometry of [Ni(CO)]4 is? - askIITians The geometry of [Ni(CO)]4 is? - askIITians](https://files.askiitians.com/cdn1/cms-content/common/www.askiitians.comonlinetestforumsimages261-2458_3449166.png.jpg)
![Draw the structure of: (i) [Ni(CO)(4)] (ii) [Fe(H(2)O)(6)]^(+3) Draw the structure of: (i) [Ni(CO)(4)] (ii) [Fe(H(2)O)(6)]^(+3)](https://d10lpgp6xz60nq.cloudfront.net/physics_images/DBT_SM_CHE_XII_U_08_E03_004_S01.png)